Nanoengineered transition metal oxygen electrodes may be able to boost lithium oxygen battery current output by orders of magnitude and extend electric vehicle ranges to 500 miles.
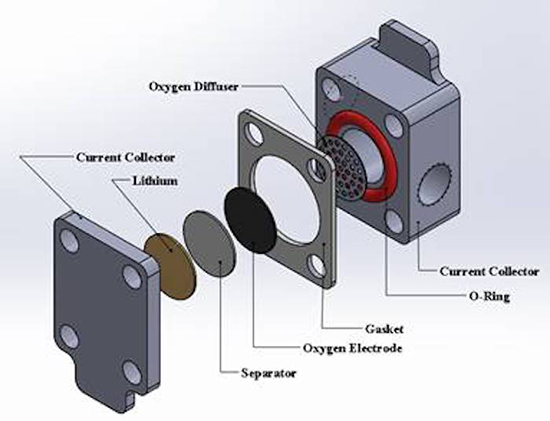
Electric vehicles use lithium ion batteries and have ranges up to 300 miles, but a lithium oxygen battery could be 75 percent lighter and allow cars to travel for up to 500 miles. A battery works with a cathode electrode and an anode electrode and an electrolyte that transfers electrons between them. In a lithium oxygen battery, the cathode is the oxygen from the air. This is why the battery is so much lighter, the cathode is absorbed from the atmosphere.
“The battery size is only one third or a quarter of existing batteries. So, it is going to be much cheaper. With less material, it is going to much cheaper,” says Xianglin Li, assistant professor of mechanical engineering at the University of Kansas. Li has been working on lithium oxygen batteries since 2014. But, the drawback is lithium oxygen can only deliver milliamps per square centimetre of electrode, while a hydrogen powered fuel cell, for example, can provide one amp.
“The [lithium oxygen] current is extremely low, compared with other lithium-ion batteries or a fuel cell, three orders of magnitude lower and that limits the power because the current multiplied by the voltage is the power,” says Li. The solution to the low current problem is an oxygen electrode that is designed with a much higher surface area for the absorption of oxygen from the ambient atmosphere.
This porous electrode for the oxygen will be nanoengineered and structured to maximise its surface area. The researchers are also aiming to understand how the oxygen is transported according to the pore size and their structure.
Catalysts are also important to the oxygen cathode process. Carbon has been used as a catalyst for lithium oxygen batteries, but the performance is inadequate. Li explains that his collaborators have identified transition metals as a catalyst candidate. A transition metal is a metallic element from one part of the periodic table, and they include, iron, manganese, chromium and copper. The transition metal catalysts are better for battery charging and discharging. “We want fast as possible catalyst reactions,” Li adds.
To analyse the performance of this electrode and battery, Li and his collaborators will use the X-ray Computed Tomography Facility at Carnegie Mellon University. Computed tomographic (CT) three-dimensional imaging involves firing X-rays at an object from multiple orientations and then a computer builds up a picture of the internal structure of the target. This CT process will allow the researchers to examine the electrode down to a scale of 15 nanometres.
Li explains that there are competing technologies. They are zinc-air or lithium-sulphur batteries, but he says that lithium-oxygen has the highest energy capacity. Li has received a $219,312, two-year grant from the United States’ National Science Foundation and support from his own Kansas University.
