Argonne National Laboratory has undertaken a study of the basic mechanisms that allow electrolyte additives to dramatically improve the performance and cycle-life of lithium ion batteries.
Battery researchers have discovered one reason why lithium ion batteries gradually lose their ability to store energy: a degradation of the electrolyte due to interactions with the cathode materials. Recent research is adding to an understanding of why this degradation occurs and how it can be controlled through the addition of additives to the electrolyte.
Batteries are made up of an anode (negative electrode) and a cathode (positive electrode) separated by an electrolyte through which ions can flow. In a lithium ion battery, that anode is usually a carbon graphite material that can accept and store lithium ions between its layers of carbon atoms. The cathode is typically a metal oxide composed of nickel, manganese, cobalt, and lithium. It is the cathode that limits the maximum storage capacity of a lithium ion battery.
The typical electrolyte for a lithium ion battery is a lithium salt (typically lithium hexafluorophosphate, LiPF6) mixed into an organic solvent. That solvent is often a mix of ethylene carbonate, dimethyl carbonate, propylene carbonate, dimethyl carbonate, and ethyl methyl carbonate. The electrolyte must allow the passage of lithium ions between the electrodes during charging and discharging. It must also prevent the passage of electrons to avoid self-discharging of the battery cell.
Electrolytes must not react with either the anode or cathode materials or the result will be a gradual degradation in battery performance. This is not a problem for the carbon of the anode, but the metal oxides in the cathode present a problem. When the cathode is in contact with the electrolyte over a long period of time, the liquid electrolyte begins to degrade, reducing battery performance.
Boosting Performance
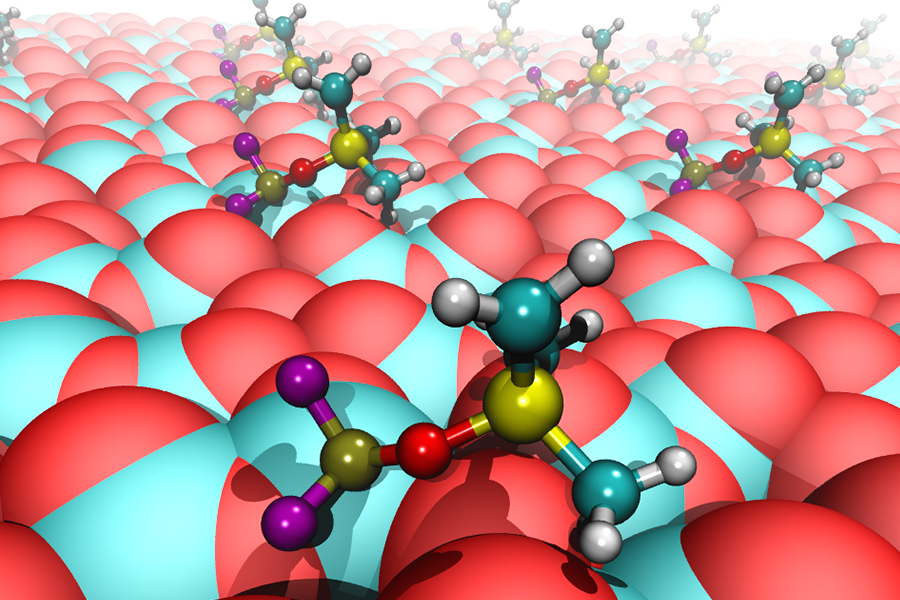
The good news is that a performance-boosting additive can be added to the electrolyte. The material is called tris(trimethylsilyl)phosphite, or TMSPi. According to a press release from Argonne National Laboratory, which has studied TMSPi in lithium ion batteries, the additive modifies the cathode surface. In doing so, it forms a protective layer to help reduce the degradation of the electrolyte near the cathode surface.
According to Argonne senior materials scientist Daniel Abraham, the mechanism by which TMSPi works in the lithium ion battery was a mystery—until now. A recently published article in The Journal of Physical Chemistry describes work undertaken by Abraham and Argonne to understand how the additive provides protection that enhances battery performance.
According to the Argonne press release, what is surprising about the latest research is the discovery that the TMSPi molecule itself is not involved directly in the protection of the cathode. The release reports, “The active component is a different molecule, PF2OSiMe3, which is chemically derived from TMSPi.” (The “Me” in this formula represents a methyl group.) The compound slowly forms as the lithium salt in the electrolyte reacts with the TMSPi.
Benefits
Abraham calls the process “pickling” and notes that it has several beneficial effects. First, the reaction product reduces the rise in electrical resistance that normally occurs after many charge and discharge cycles. Slowing of the lithium ions traveling between the cathode and anode and a change in the cathode chemistry due to an interaction with the electrolyte are the cause of this rise in resistance. The new active compound decreases the rise while allowing faster charging and discharging of the lithium ion cells.
Another beneficial effect of the TMSPi-derived molecule is that it reduces the loss of cobalt and manganese transition metals from the cathode into the electrolyte and eventually reaching the anode. The result of the loss and transfer can be a degradation of performance after many cycles. This is prevented by the presence of the active molecule.
Physical Understanding
While performance improvements were straightforward to measure, an actual physical understanding of the mechanisms involved required more work. A computational study at Argonne revealed that the reaction product PF2OSiMe3 binds to reaction centers on the surface of the cathode without removing oxygen from the surface. According to the release, “This surface-bound molecule can further react with the electrolyte, transforming into a still-stronger binding molecule that permanently caps the reaction centers on the cathode, stabilizing the interface between liquid electrolyte and solid electrode.” Argonne’s research showed that the performance of the battery actually improves with age as more of these transformations occur.
There are important practical applications from these results. “Now that we better understand the mechanism for the cathode-protective action by the phosphite, we can be more systematic in finding new ways of achieving and improving this pickling of the electrolyte additive,” said Abraham.
